Throwing the DISE: One institution’s quest for a protocol
Myron Yaster MD and Jamie Peyton MBChB, MRCP, FRCA
Today’s Pediatric Article of the Day comes from the journal Otolaryngology–Head and Neck Surgery, a journal that I (Myron), and I’m sure many of you, have never read. This multi-disciplinary research describes a quality improvement project designed to develop a better and more consistent anesthetic plan for drug induced sleep endoscopy (DISE). Where to publish this type of research is always problematic, an ENT journal or an anesthesiology journal? Both groups benefit from and need to know the results. Since this was published in an ENT journal, I thought it would be a good idea to share the results with you. I’ve asked Jamie Peyton MBChB, MRCP, FRCA, Assistant Professor in the Department of Anesthesiology, Critical Care and Pain Medicine at Boston Children’s Hospital to co-write this with me. Aside from lots of initials after his name and being a great guy, Jamie was the anesthesiologist involved in establishing the DISE service at Boston Children’s Hospital many years ago and has extensive experience in both anesthesia for DISE and shared airway surgery. His views on this original article will limit my personal bias. (The study was performed in Colorado where I used to work and one of the authors is my close friend. Further, all of the authors are true “believers” in the church of DISE, a church in which I am thoroughly agnostic). Myron Yaster MD
Original article
Kristin L Mooney, Melissa Brooks Peterson, Jonathan R Skirko, Norman R Friedman. The Quest for a DISE Protocol. Otolaryngol Head Neck Surg. 2021 Aug 17;1945998211036645. doi: 10.1177/01945998211036645. PMID: 34399647
“Drug-induced sleep endoscopy (DISE) is increasingly utilized by pediatric otolaryngologists to determine the anatomic site of obstruction for children with polysomnogram (PSG)–confirmed obstructive sleep apnea (OSA). This is especially true for children with persistent OSA following a tonsillectomy with or without adenoidectomy (T&A). Since the success of T&A is variable, DISE has increased in popularity as a diagnostic and planning tool. In a meta-analysis of 1079 patients, Durr et al found that at least one-third of children experienced persistent OSA following a T&A.1 The cure rate is even lower for children with Down syndrome.2 For sleep surgeons, DISE is quickly becoming what bronchoscopy is to the airway surgeon”.3
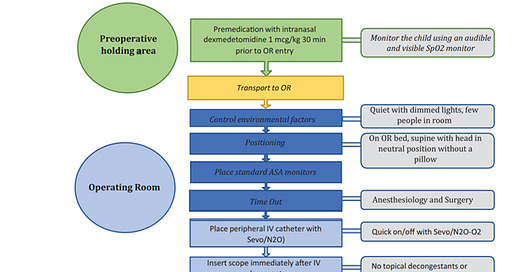
“The anesthetic and surgical goal of DISE is to choose a combination and sequence of pharmacologic agents that simulate natural sleep, are well tolerated by the child, and allow accurate identification of the obstructive breathing pattern for development of a surgical plan”.3 Although many institutions have a standard DISE protocol, none has been widely accepted. Every anesthetic used in DISE, be it dexmedetomidine, propofol, ketamine, midazolam used alone or in combination has some benefits and downsides. Thus, the authors used the QI process of plan-do-study-act (PDSA) to “perfect” a DISE anesthetic plan which in its final version is depicted in the figure below.
Ideally, dexmedetomidine should produce the desired conditions for DISE. Unfortunately -- and not surprisingly -- it fails during the 2 most stimulating steps in the DISE procedure: IV placement and insertion of the flexible scope past the posterior termination of the inferior turbinates. In this protocol, general anesthesia with N2O and sevoflurane were used for these 2 steps, and the two steps were performed nearly simultaneously. Once the scope was inserted, the vapor anesthetics were turned off (“rapid on-rapid off”) and a dexmedetomidine loading infusion 2 mcg/kg was given over 10 minutes followed by a continuous infusion of 2 mcg/kg/HOUR. Note that dexmedetomidine was also given as a premed 1 mcg/kg intranasally 30 minutes prior to going to the OR as well.
Ok, the authors created a reliable DISE protocol for their institution, but does it simulate a child’s obstructive breathing pattern during unmedicated natural sleep, which after all is the whole point? Based on this study, we don’t know because this was not part of the study design. Further, it would be difficult to make this part of the study design because “surgical outcomes are linked to the accuracy of DISE replicating a child’s true obstructive breathing pattern, a surgeon’s interpretation of DISE findings, selection of surgical treatment plan, and the technical success of the surgery. The interrater reliability of DISE is variable4 and depends on the surgeon’s experience”.5 This is why I (MY) remain a vocal non-believer in this church. Why believe the results of the DISE procedure if there is no interobserver reliability? Further, in this study neither the anesthesiologist nor the surgeon were blinded to the protocol. Did they actually achieve the conditions required to make airway evaluation valid? As they state: “Since there are no validated pediatric DISE sedation scales, the surgeon utilized the PSG asleep video description and baseline asleep SpO2 to provide guidance when a child was at the appropriate depth of sedation to assess the airway”. And as stated previously, different surgeons may have completely different views of the outcome. One interesting and useful scale the authors did choose to measure was surgeon and anesthesiologist Likert scale ratings of success of the procedure. At every PDSA cycle until the final iteration, they disagreed. With the final protocol, both surgeon and anesthesiologist concurred.
Despite my (MY) misgivings, neither Jamie nor I think DISE is going away. The members of the Society for Pediatric Anesthesia have demonstrated the value of developing multi-institutional improvement networks.6-8 Working together with our ENT colleagues, this may be an ideal area for future collaboration and study.
From my perspective (JP) I found it an interesting study to read and reflect on how the approach we have taken at BCH differs, as I did not create a departmental anesthetic protocol for these patients and it is always good to question whether decisions made several years ago (around 2013 I think) stand the test of time and new information.
Accepting that DISE is a surgical technique that can identify anatomical areas that cause upper airway obstruction and that may be amenable to surgical correction is the first step. I think that the data we have does support this, in particular it appears useful in children who have already had a tonsillectomy +/- adenoidectomy and still have significant OSA.
There is some evidence that DISE guided surgery improves OSA in the short term, but we lack long-term results. There is no currently available evidence that the anesthetic technique used during the DISE makes any difference to the choice of operation, and therefore the subsequent outcome of any surgery. The concerns that some drugs (particularly volatile agents) will cause too much of a decrease in neuromuscular tone giving false results is mirrored by concerns that other drugs (mainly ketamine) may cause an increase in neuromuscular tone and not cause appropriate airway obstruction, thus leading to an under appreciation of the degree of airway obstruction that is occurring during sleep. This has led to a drive to use single agents, notably dexmedetomidine as it is thought to mimic natural sleep more closely than any other anesthetic agent. When compared to propofol, the loss of airway tone and airway collapse is similar at the same levels of sedation created by both drugs. When you look more closely at dexmedetomidine (the drug of choice in the paper we are discussing), it seems to alter sleep architecture by decreasing the proportion of REM to NREM sleep. OSA is predominantly a REM phenomenon in children with an increase in upper airway collapse associated with the withdrawal of excitatory noradrenergic and serotonergic inputs to upper airway motor neurons during REM sleep. This implies that dexmedetomidine is not without potential issues when it comes to the claim that it produces the correct conditions to assess ‘natural’ upper airway collapse. Indeed, a recent study trying to ascertain if any difference in airway obstruction occurred when propofol was used instead of dexmedetomidine during DISE showed that there was no difference in the degree of upper airway obstruction produced by either drug.9
The emphasis on protocol driven surgery and anesthesia has been largely driven by the surgical teams involved in developing DISE. Perhaps the clearest recent example of this lies in the expert consensus statement published in January 2021,10 stating that there is, “an opportunity for quality improvement regarding the sedation for pediatric DISE since there is significant practice variation in this area”. No anesthesiologists are authors on this expert consensus statement, or are recorded in the methods as having been asked their opinion on the anesthetic management of these cases. All the authors are ENT surgeons! Our ENT surgical colleagues have a huge amount of expertise in this field, but they are not anesthesiologists; their work would benefit from the ego-aside, true multidisciplinary involvement of those responsible for choosing which medications to administer. This is especially true when there is a concern that the drugs used to induce OSA-like airway collapse will change the outcome of their decision-making during the DISE and therefore the ultimate surgical procedure. Could you imagine, even for a minute, how cranky these surgeons would be if anesthesiologists created a consensus position paper on DISE without surgical participation in the drafting and writing of the paper? In a recent PAAD, August 16, Authorship: Credit and Credibility, I (MY) discussed some of these issues, but I must admit that it pisses me off that surgeons would write and publish a “consensus” paper about anesthetic management without having their anesthesia colleagues acknowledged in the authorship choo-choo train. But I digress…
The reason I (JP) did not create a protocol for DISE in my hospital was that the patients I was being asked to anesthetize for the procedure were so heterogenous and complex that a ‘one-size-fits-all’ approach was impossible. The anesthetic I would give to an otherwise healthy, if slightly obese 13 year old differs from the 6 year old with a Fontan circulation, or the 12 year old with trisomy 21 and supra-systemic RV pressures, or the infant with Robin sequence. The key for me was that the surgeon required a patient breathing spontaneously at varying depths of anesthesia, depending upon which part of the process he was doing, and that this was achievable by many different techniques. Clear communication and an efficient, systematic approach to the DISE during periods where airway obstruction is deliberately induced by the anesthesia team is more important than how we induced airway obstruction.
What would be interesting is to look at the patients who have undergone DISE in institutions where the anesthetic is protocol-based, and compare the results of the DISE, the surgical choices, and the outcome of the surgery with patients from institutions where the anesthetic choice is left up to the individual anesthesiologist. Obviously, this would require consensus and inter-observer reliability which currently does not appear to exist.
Myron Yaster MD and Jamie Peyton MBChB, MRCP, FRCA
References
1. Durr ML, Meyer AK, Kezirian EJ, Rosbe KW: Drug-induced sleep endoscopy in persistent pediatric sleep-disordered breathing after adenotonsillectomy. Arch Otolaryngol Head Neck Surg 2012; 138: 638-43
2. Ingram DG, Ruiz AG, Gao D, Friedman NR: Success of Tonsillectomy for Obstructive Sleep Apnea in Children With Down Syndrome. J Clin Sleep Med 2017; 13: 975-980
3. Mooney KL, Brooks Peterson M, Skirko JR, Friedman NR: The Quest for a DISE Protocol. Otolaryngol Head Neck Surg 2021: 1945998211036645
4. Altintaş A, Yegin Y, Çelik M, Kaya KH, Koç AK, Kayhan FT: Interobserver Consistency of Drug-Induced Sleep Endoscopy in Diagnosing Obstructive Sleep Apnea Using a VOTE Classification System. J Craniofac Surg 2018; 29: e140-e143
5. Vroegop AV, Vanderveken OM, Wouters K, Hamans E, Dieltjens M, Michels NR, Hohenhorst W, Kezirian EJ, Kotecha BT, de Vries N, Braem MJ, Van de Heyning PH: Observer variation in drug-induced sleep endoscopy: experienced versus nonexperienced ear, nose, and throat surgeons. Sleep 2013; 36: 947-53
6. Uhl KM, Wilder RT, Fernandez A, Huang H, Muhly WT, Zurakowski D, Cravero JP: Postoperative pain and psychological outcomes following minimally invasive pectus excavatum repair: A report from the Society for Pediatric Anesthesia Improvement Network. Paediatr Anaesth 2020; 30: 1006-1012
7. Garcia-Marcinkiewicz AG, Kovatsis PG, Hunyady AI, Olomu PN, Zhang B, Sathyamoorthy M, Gonzalez A, Kanmanthreddy S, Gálvez JA, Franz AM, Peyton J, Park R, Kiss EE, Sommerfield D, Griffis H, Nishisaki A, von Ungern-Sternberg BS, Nadkarni VM, McGowan FX, Jr., Fiadjoe JE: First-attempt success rate of video laryngoscopy in small infants (VISI): a multicentre, randomised controlled trial. Lancet 2020; 396: 1905-1913
8. Goobie SM, Cladis FP, Glover CD, Huang H, Reddy SK, Fernandez AM, Zurakowski D, Stricker PA, Gries H: Safety of antifibrinolytics in cranial vault reconstructive surgery: a report from the pediatric craniofacial collaborative group. Paediatr Anaesth 2017; 27: 271-281
9. Kirkham EM, Hoi K, Melendez JB, Henderson LM, Leis AM, Puglia MP, 2nd, Chervin RD: Propofol versus dexmedetomidine during drug-induced sleep endoscopy (DISE) for pediatric obstructive sleep apnea. Sleep Breath 2021; 25: 757-765
10. Baldassari CM, Lam DJ, Ishman SL, Chernobilsky B, Friedman NR, Giordano T, Lawlor C, Mitchell RB, Nardone H, Ruda J, Zalzal H, Deneal A, Dhepyasuwan N, Rosenfeld RM: Expert Consensus Statement: Pediatric Drug-Induced Sleep Endoscopy. Otolaryngol Head Neck Surg 2021: 194599820985000




Very interesting 🤔