Remembering the Classics: Changes in serial platelet counts following massive blood transfusion in pediatric patients
Charles J. Coté MD
As regular readers of the PAAD know, I, like the late Ron Litman before me, often like to revisit classic articles and ask the original authors to comment not only on their findings but why they did their study and the obstacles they had to overcome in order to perform their studies. Having recently posted a PAAD on pediatric blood management (July 3, 2025: https://ronlitman.substack.com/p/pediatric-patient-blood-management ) I thought it would be an ideal time to revisit a classic paper on pediatric transfusion therapy. I asked its lead author of today’s PAAD, Dr. Charles (Charlie) Coté, to “talk story”. Myron Yaster MD
Original article
Coté CJ, Liu LM, Szyfelbein SK, Goudsouzian NG, Daniels AL. Changes in serial platelet counts following massive blood transfusion in pediatric patients. Anesthesiology. 1985 Feb;62(2):197-201. doi: 10.1097/00000542-198502000-00024. PMID: 3970376.
I read with great interest the PAAD on pediatric blood management and want to thank Myron for giving me this platform to add a few more thoughts and perspectives on this topic. I agree that there is precious little information regarding massive blood transfusion (MBT) in children since fortunately in the USA, we are not in a wartime setting and that gunshot injuries or MVAs resulting in the need for massive transfusion are relatively rare for each of us. At my age I can look back at the surgical practices pre HIV where oftentimes attention to blood conservation was, let’s just say, less than stellar. I was interested in this topic because of my care of children with extensive burn injuries at the Boston Shriners Burn Institute, especially with one particular surgeon, who I will call Dr. Q. On his operating days it was common to loose 1-3 blood volumes and it occurred 1-2 times per week during excision and grafting procedures! This was terrible for the patient, but it provided a research opportunity that allowed me to study massive blood transfusions in a relatively predictable way. The impetus for these studies was that the very first burn patient that I ever cared…an ~8 kg infant with a large burn injury. I started the case and went to my other room to start another plastic surgery case. My resident had been warned about Dr. Q, and being a good resident, he administered a unit of FFP over about 3 minutes to volume load the patient before blood loss even began. Within minutes I was emergently called back to the room because the infant was in EMD, and they were doing chest compressions! This happened before Dr. Q had even touched the patient. We gave several rounds of exogenous calcium and the infant rapidly and uneventfully recovered. I wondered why did this happen? Was it the citrate in FFP binding calcium, deep halothane anesthesia, or a combination of both?
After obtaining investigational review board (IRB) approval, I began my research of FFP induced citrate toxicity using controlled rates of administration. The IRB allowed me to study this in measured steps starting with the lowest proposed rate and then, after not observing severe adverse responses, they allowed me to examine 3 other rates of infusion. I ultimately studied 27 FFP infusions in children with fixed rates of administration of 1.0, 1.5, 2.0, and 2.5 mL/kg x 5 minutes (the latter equivalent to a 70 kg adult receiving ~175 mL/min or 875 mL over 5 minutes). We used a series of four way stop cocks, large glass syringes, and a four-barrel Harvard syringe pump (likely a pump that would not meet current safety standards since there was no pressure alarm!). FFP infusions were initiated at the onset of tangential excisions which with Dr. Q always resulted in a blood bath. What we found was clinically important rapid reductions in ionized calcium (but no adverse hemodynamic responses). Rates as slow as 1 mL/kg/min reduced ionized calcium from ~1.2 to ~1.0 mmol/L and rates of ≥1.5 mL/kg/min reduced ionized calcium to as low as 0.7 mmol/L.
The interesting thing that surprised me was that within 1 minute of completing the infusions, the ionized calcium values rapidly began to normalize so that by 5 minutes after the infusions the ionized calcium values had returned to ~1 mmol/L without the need for exogenous calcium. The most likely reason for this was rapid metabolism of citrate in the Kreb’s cycle releasing the bound calcium. Of note, a burn victim would be able to metabolize citrate at a far greater rate than a non-burned child due to the hypermetabolism of burns and the persistent high cardiac output in acute burn patients (more blood delivered to the liver allowing greater metabolism of citrate). I would expect that the changes in ionized calcium associated with these rates of administration would be greater in a non-burn child. Indeed, we tried to repeat the study in non-burn patients, but HIV came along and the surgeons started paying much closer attention to blood loss. We had to terminate the study (good for the children but bad luck for my research). The lesson here was that the danger of citrate toxicity is during not after a rapid FFP transfusion.[1]
Now imagine a premature 1.4 kg neonate with NEC that needs FFP, is it really possible to give that infant <1.5 mL/kg/minute without infusing the FFP on a pump? Also consider that these premies and even toddlers would have a much lower ability to metabolize citrate than my older burn patients. So, the patients at greatest risk for citrate toxicity are neonates and toddlers due to their size. the ease of rapidly infusing FFP, and immature hepatic metabolic processes. Thus, another lesson from this study: Give calcium through a separate line during the rapid infusion of citrate containing blood products. Also remember that calcium chloride and calcium gluconate ionize equally rapidly and produce equal increases when administered in appropriate doses, e.g., 3 time more mg/kg of gluconate than chloride so 15 mg/kg calcium gluconate is equivalent to 5 mg/kg calcium chloride.[2]
As part of this journey, I reviewed all intraoperative cardiac arrests at the Shriners Burns Institute for the prior 10 years and found that all had been receiving a halothane anesthetic. So I conducted a laboratory animal model study whereby I compared two situations: citrate infusions during a deep and a light plane of halothane anesthesia. We found that with deep halothane anesthesia there was significantly greater cardiac dysfunction resulting in cardiac arrest and a greater reduction in ionized calcium compared with a lighter depth of halothane anesthesia. Presumably this was due to reduced hepatic blood flow and reduced citrate metabolism during deeper planes of anesthesia[3] (this was presented in more detail in a prior PAAD discussion Feb 7, 2024). We demonstrated that the combination of inhalation agent induced cardiac depression by their calcium channel blocking activity properties, combined with a sudden fall in ionized calcium by rapid FFP administration = a disaster waiting to happen.
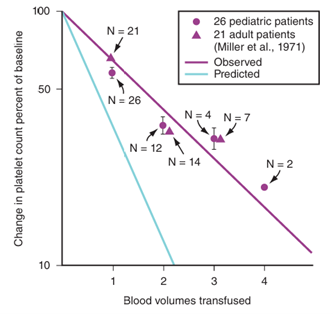
This next led me to examine serial changes in platelet counts and changes in PT/PTT with massive blood loss. I compared the serial changes in platelet counts in 26 children who lost 1-5 blood volumes. The changes we observed[4] were nearly identical to those made by Ron Miller (figure 1) during his experience in Vietnam. [5, 6]
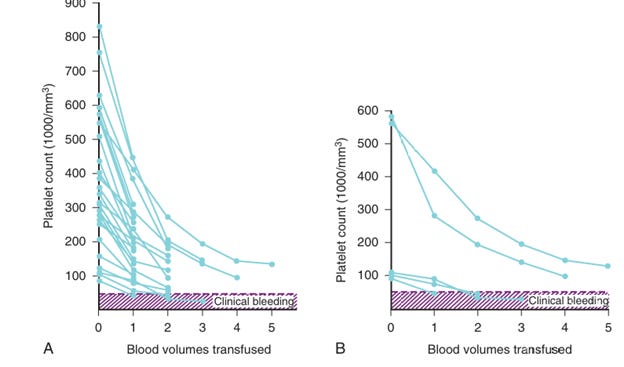
We observed that on average the platelet count was reduced by ~40% from baseline at 1 blood volume loss, another 30% after the second and a further 10% after the third. Thus, after losing 3 blood volumes one can expect an approximate 70% reduction from baseline. We observed three children who developed clinical bleeding when the platelet count was <50,000/mm3 and all started with platelet counts of ~100,000/mm3 since their baseline platelet counts were very elevated. We had two others who lost 4 and 5 blood volumes who did not require platelets since even with this large blood loss their platelet counts remained above 100,000/mm3(Figure 2). Left is all 26 patients and Right = five illustrative cases).
The lesson here is that with relatively controlled blood loss if you know the baseline platelet count you can estimate when exogenous platelets might be required and the 1:1:1 rule may not apply despite massive blood loss.
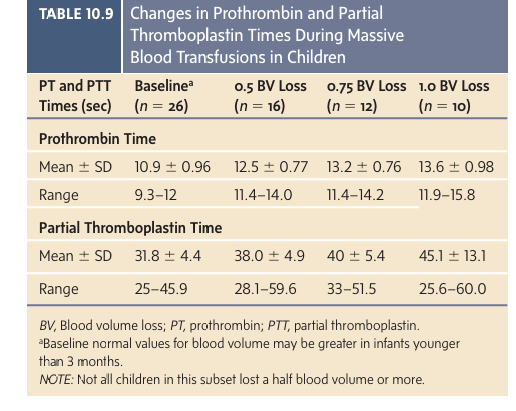
We also looked at changes in prothrombin and partial thromboplastin times (PT/PTT) at 1, 1.5, and 2.0 blood volumes lost in ten children when FFP was not replaced (FFP not available and hemorrhage unexpected) (we stopped at 10 for the same reason we stopped the FFP study described above; we had no patients in the following year loose this much blood so the study was closed early). In those 10 children we observed minimal changes in either PT or PTT at one blood volume blood loss but significant prolongations after this point. So, the break point for needing FFP seems to be after one blood volume has been shed and replaced (Table). Recall that the labile clotting factors V and VIII in whole blood are still within 20-50% of baseline so using whole blood rather than PRBCs would likely eliminate the need for FFP in most cases (if it were available in large quantities as it was for Dr. Miller in Vietnam). We have come full circle on this issue; whole blood would in fact be better than component therapy in a MBT situation.
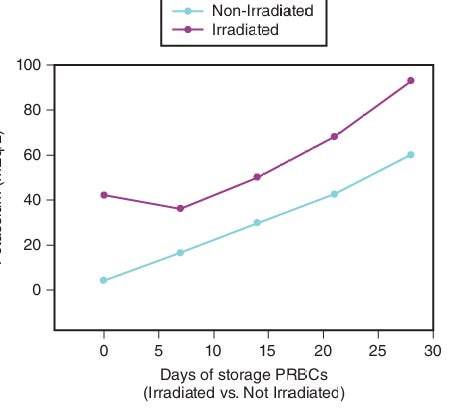
Now this brings us to MBT protocols recommending 2:1:1 or 1:1:1 PRBCs, FFP, and platelets. Way back in the early editions of my book (Editions 2 and 3) I recommended that, based on Miller’s published studies, one needs >30% of factor VIII and 20% of factor V to have normal PT and PTT values.[5, 6] Based on Miller’s data in adults and my pediatric data, I recommended that platelet transfusions should be based on measured platelet counts. I also admonished that one should anticipate clinically important changes in the PT/PTT after one blood volume is lost and replaced with PRBCs. I suggested that after one blood volume is shed, for each blood volume thereafter this replacement should consist of 1/3 blood volume replaced with PRBCs, 1/3 with FFP and 1/3 with other fluids (albumen, LR, etc., NOT PLATELETS); 1/3 blood volume replacement with FFP would maintain the labile factors V and VIII withing the clotting range. Looking back now with the more recent trauma recommendations, my observations and recommendations for pediatric patients were quite similar to current adult MBT protocol recommendations. Obviously, the difference for children is we cannot talk about units of blood transfused since a unit of PRBCs given to a robust 100 kg teenager would be a fraction of a blood volume but, given to a 1.4 kg premie, it would be multiple blood volumes. So we have to clearly quantitate the volumes of each blood product administered as they relate to the child’s estimated blood volume and losses. One additional concern is that stored blood has increasing levels of potassium (Figure 3) as storage time extends such that rapid and large volume administration (especially older blood) may result in hyperkalemic cardiac arrest, which is less likely if the blood is transfused peripherally rather than centrally.[7] [8]
So, my recommendations are 1 PRBCs:1 FFP:1 other in ratios of blood volumes not units (try to use washed PRBCs in premies and neonates if rapid administration is anticipated). In part this approach relates to my residency when one of my attendings, D Eric Greenhow, preferred to use the phrase “1 Harvey = 10% of a blood volume” when comparing the coagulation and complications of transfusions in an elderly debilitated female weighing 45 kg vs an elderly obese male weighing 90 kg. Harveys still make more sense for children than units, especially for neonates and infants; think Harveys not units!
All figures and the table are from Coté CJ, Grabowski EF, Sloan SR, Strategies for Blood Product Management, Reducing Transfusions, and Massive Blood Transfusion, Chapter 10, in A Practice of Anesthesia for Infants and Children 7th Edition Elsevier 2025
Send your thoughts and comments to Myron who will post in a Friday reader response.
References
1. Coté CJ, Drop LJ, Hoaglin DC, Daniels AL, Young ET: Ionized hypocalcemia after fresh frozen plasma administration to thermally injured children: effects of infusion rate, duration, and treatment with calcium chloride. Anesth Analg 1988, 67(2):152–160.
2. Cote CJ, Drop LJ, Daniels AL, Hoaglin DC: Calcium chloride versus calcium gluconate: comparison of ionization and cardiovascular effects in children and dogs. Anesthesiology 1987, 66(4):465–470.
3. CJ C: Depth of halothane anesthesia potentiates citrate-induced ionized hypocalcemia and adverse cardiovascular events in dogs. Anesthesiology 1987, 67:676–676.
4. Coté CJ, Liu LM, Szyfelbein SK, Goudsouzian NG, Daniels AL: Changes in serial platelet counts following massive blood transfusion in pediatric patients. Anesthesiology 1985, 62(2):197–201.
5. Miller RD, Robbins TO, Tong MJ, Barton SL: Coagulation defects associated with massive blood transfusions. Annals of surgery 1971, 174(5):794–801.
6. Miller RD: Complications of massive blood transfusion. Anesthesiology 1973, 39:82–93.
7. Lee AC, Reduque LL, Luban NL, Ness PM, Anton B, Heitmiller ES: Transfusion-associated hyperkalemic cardiac arrest in pediatric patients receiving massive transfusion. Transfusion 2014, 54(1):244–254.
8. Burke M, Sinha P, Luban NLC, Posnack NG: Transfusion-Associated Hyperkalemic Cardiac Arrest in Neonatal, Infant, and Pediatric Patients. Front Pediatr 2021, 9:765306.