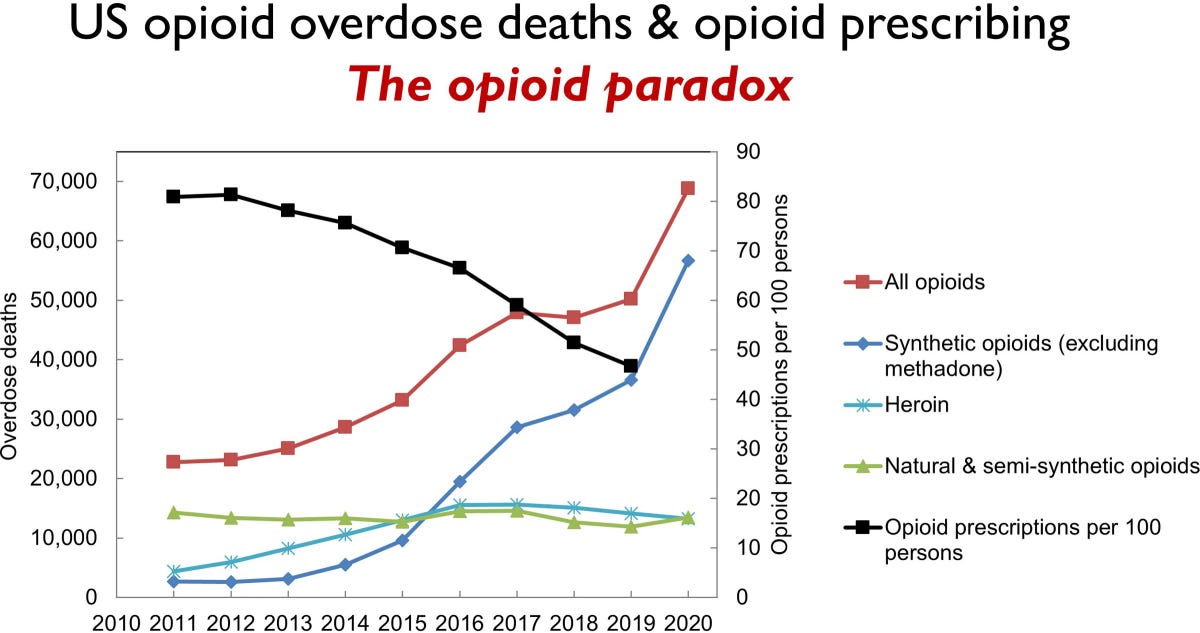
This is hard to believe…during the Covid-19 pandemic opioid overdose deaths actually spiked to record new highs … an epidemic within a pandemic. Opioids now account for 74% of all fatal drug overdoses in the United States reaching 66,000 deaths, or 181 deaths/day or 1 death every 8 minutes! But unlike most of the 2000’s and 2010’s, now the main culprit is illegal/street fentanyl, which today accounts for two-thirds of all opioid overdose deaths. Indeed, this increasing opioid overdose death toll has occurred despite steady reductions in opioid prescribing by physicians, the so called “opioid paradox” (see figure at the bottom of the PAAD). As the authors of today’s special article point out “it is abundantly clear that current approaches to the opioid crisis are not succeeding”. I’ve asked Elliot Krane to co-write this PAAD with me. Elliot and his colleagues Steve Weisman and Gary Walco predicted this “paradox” of increasing opioid deaths despite prescription opioid reduction shortly after the CDC put into place recommendations for restrictions in opioid prescribing,(1) which were then enthusiastically established by state legislatures and medical societies, sometimes with Draconian modification. They also warned that a one size fits all mandate for how much opioid could be dispensed would invariably result in undertreatment of pain. The late Ron Litman, who started the PAAD, wrote that abuse deterrent opioid products reduced opioid abuse but had little to no effect on opioid overdose death rates.(2) Indeed, the real paradox is that by reducing the abuse potential of sustained release products by adding tamper resistant components and limiting how much opioid could be dispensed resulted in opioid abusers being driven to the cheaper and more available substitute for oxycodone: heroin. Just a few years later, supply and demand economics further shifted street inventory to fentanyl as a cheaper, non-seasonal, easier to manufacture and far, far easier to smuggle substitute for heroin, with catastrophic consequences. In today’s PAAD we will review some of the highlights of this paper.
Special article
Evan D Kharasch, J David Clark, Jerome M Adams. Opioids and Public Health: The Prescription Opioid Ecosystem and Need for Improved Management. Anesthesiology. 2022 Jan 1;136(1):10-30. PMID: 34874401
The authors introduce the concept of the “prescription opioid ecosystem:” The pool of unused prescription opioids available for diversion, misuse, and overdose. This pool is influenced by supply, demand, and return or disposal. Over-prescription of opioids, while providing sufficient supply to treat the needs of those with pain, risks potential overuse of residual unused opioids, while under-prescription risks undertreating acute and chronic pain. Yet reducing supply alone has not been successful at reducing opioid overdose rates. Reducing opioid need or demand through better pain management, while enhancing disposal and return, are additional approaches to reducing the opioid pool and mitigating opioid misuse, abuse and accidental overdose deaths”.
Let’s start with demand. Acute and chronic pain are very common. In today’s PAAD, we are going to concentrate on postoperative pain and inter- and intra-patient variability. How much pain patients experience and their response to opioids are highly variable. A one size fits all method for how much opioid to prescribe will invariably result in too much medication dispensed to some and not enough to others.
Supply. The variability in patient response is not limited to patients it is also influenced by the prescriber. How much opioid is dispensed is influenced by years of practice, type of surgery, provider desire to minimize prescriber inconvenience (refills), prescribing guidelines, provider knowledge, lack of knowledge, bias and paranoia, and even by local politics and culture.(3-5) Unfortunately “…the nation’s response to the oral opioid crisis has been to tighten patient supplies and impose institutional and practitioner quality indicators based on pill counts. Governments, payers, and pharmacies have assumed authority for limiting opioid prescribing, often in indiscriminate ways.” “Pill counts have become de facto standards employed by healthcare organizations to highlight their success in reducing opioid use, yet there is no discussion of how those reductions are affecting patient outcomes. One crucial problem is that agencies mandating policy restrictions do not measure, nor are they accountable for, patient outcomes.”(1) A policy of limiting the supply of prescription opioids to eliminate opioid abuse deaths seems logical on its face but is analogous to limiting the supply of gasoline to eliminate drunk driving deaths. The desired effect may occur, but it is uncertain. What is certain is that such a policy will cause significant collateral damage.
The opioid left over pool. Left over opioids represents a pool available for potential diversion, misuse, addiction, and accidental overdose. Multiple studies have shown that most patients do not and will not return or dispose of unused prescription opioids, and yet do not store them in locked or inaccessible locations.(6-8) Children and adolescents comprise a vulnerable population affected by the unused opioid pool. Adolescent opioid misuse is associated with subsequent dependence, opioid use disorders, heroin use, and overdose(9), although the causal link is a matter of debate. In other words, it is not clear whether the misuse and abuse of unused opioids is a phenomenon largely restricted to teens who are otherwise experimenting with abusable substances and who are already at risk for substance use disorders, or whether it traps otherwise low risk innocent teens in a cycle of misuse, abuse and dependence. Most evidence points to the former.
Managing the pool: Disposal. We think there will always be left over, unconsumed opioids. Thus, how to manage this pool is a key in the opioid ecosystem. Most people keep their unused opioids for potential future need, and disposal methods are inconvenient, confusing, or may not actually work.(10) One new idea raised by the Kharasch et al., and recently enacted into law, is for partial filling of opioid prescriptions. Currently, if a prescriber orders 30 tablets of a drug, the pharmacist must dispense all 30. What if they could dispense only some of the drug AND if the patient needed more they could get it at the pharmacy without a new prescription and without another co-pay?
One final thought. Not discussed by the authors in today’s PAAD is the dispensing of intranasal naloxone when prescribing opioids. We discussed intranasal naloxone in previous PAAD (July 26,2021). Intranasal naloxone is a life-saving treatment that we need to think about whenever we send patients home with opioids. As one of the authors of today’s paper, the former surgeon general of the United States Dr. Jerome M Adams pointed out at a past ASA annual meeting, we spend so much effort on CPR training, shouldn’t we be concentrating on opioid overdose deaths as well and give everyone nasal naloxone for emergency resuscitation use? And if not everyone, we believe that nasal naloxone should be co-dispensed with opioid prescriptions at least in high risk circumstances. High risk situations could include co-prescription of benzodiazepines or other sedatives, and/or prescription of high dose opioids (>100 MED/24hr), methadone, sustained release opioids, oral transmucosal and transdermal opioids, as well as opioid prescription in high risk medical and social circumstances including to households with small children, and to patients with obesity or OSA, and with a history or family history of substance use disorders or psychiatric diagnoses associated with substance abuse.
Myron Yaster MD and Elliot J. Krane MD
References
1. Krane EJ, Weisman SJ, Walco GA. The National Opioid Epidemic and the Risk of Outpatient Opioids in Children. Pediatrics 2018;142.
2. Litman RS, Pagan OH, Cicero TJ. Abuse-deterrent Opioid Formulations. Anesthesiology 2018;128:1015-26.
3. Santosa KB, Wang CS, Hu HM, Brummett CM, Englesbe MJ, Waljee JF. Surgeon experience and opioid prescribing. Am J Surg 2020;220:823-7.
4. Kattail D, Hsu A, Yaster M, Vozzo PT, Gao S, Thompson JM, Roter DL, Laporte D, Henn RF, 3rd, Fiadjoe JE, Monitto CL. Attitudes and self-reported practices of orthopedic providers regarding prescription opioid use. Journal of opioid management 2019;15:213-28.
5. Hunsberger JB, Monitto CL, Hsu A, Yenokyan G, Jelin E. Pediatric surgeon opioid prescribing behavior: A survey of the American Pediatric Surgery Association membership. J Pediatr Surg 2021;56:875-82.
6. Bicket MC, Long JJ, Pronovost PJ, Alexander GC, Wu CL. Prescription Opioid Analgesics Commonly Unused After Surgery: A Systematic Review. JAMA surgery 2017.
7. Hunsberger JB, Hsu A, Yaster M, Vozzo PT, Gao S, White ED, Yenokyan G, Vickers B, Monitto CL. Physicians Prescribe More Opioid Than Needed to Treat Pain in Children After Outpatient Urological Procedures: An Observational Cohort Study. Anesth Analg 2019.
8. Monitto CL, Hsu A, Gao S, Vozzo PT, Park PS, Roter D, Yenokyan G, White ED, Kattail D, Edgeworth AE, Vasquenza KJ, Atwater SE, Shay JE, George JA, Vickers BA, Kost-Byerly S, Lee BH, Yaster M. Opioid Prescribing for the Treatment of Acute Pain in Children on Hospital Discharge. Anesth Analg 2017;125:2113-22.
9. Kelley-Quon LI, Cho J, Strong DR, Miech RA, Barrington-Trimis JL, Kechter A, Leventhal AM. Association of Nonmedical Prescription Opioid Use With Subsequent Heroin Use Initiation in Adolescents. JAMA pediatrics 2019:e191750.
10. San Francisco Department of the Environment. Medicine disposal products an overview of products & performance questions, 2019.
Figure 1
Figure 2