Reader response
Myron Yaster MD
From Jeffrey Feldman, MD, MSE, FASA, Children’s Hospital of Philadelphia (retired)
I read the PAAD by Rowland et al published on May 7 with interest here. The findings they describe from the article by Tian et al are not at all surprising. A number of studies published beginning in the early 1990s by authors under the direction of Goran Hedenstierna clearly showed that 1) 100% oxygen causes atelectasis, 2) atelectasis does not occur if the oxygen concentration is limited to less than 80% and 3) recruitment maneuvers are effective for reversing atelectasis but need to be followed by limited oxygen concentration and PEEP to prevent atelectasis formation. The figure is from a study by Rothen et al published in 1995 that provides some insight into how rapidly atelectasis forms in the presence of 100% oxygen. These studies are quite compelling in that atelectasis was measured by computed tomography and often included data documenting impaired oxygenation and reduced lung compliance. Since this is not a review article format, I will focus on a few studies in particular that specifically addressed questions relevant to this discussion:
· What is the optimal oxygen concentration during induction of general anesthesia? This question was actually the title of an article by Edmark et al reporting on the use of three concentrations of oxygen during induction of 36 adult women – 100, 80 and 60 percent. Atelectasis formation documented by CT showed minimal atelectasis in the 60% and 80% groups compared with the 100% group. Of note, the apnea time to a desaturation of 90% was longer in the 100% group at 411 secs average v 303 and 213 secs in the 80% and 60% groups respectively.
Edmark et. al. Anesthesiology. 2003;98:28.
· What is the optimal oxygen concentration to use during emergence from anesthesia? Since 100% oxygen causes atelectasis, what is the risk for postoperative atelectasis if 100% oxygen is used during emergence. This question was investigated by Benoit et al in 30 adult patients who were randomized 10 minutes before anticipated extubation to 100% oxygen, a vital capacity maneuver then 100% oxygen or a vital capacity maneuver then 40% oxygen. Immediately after extubation, patients were transported for thoracic CT to measure atelectasis. Both groups with 100% oxygen had substantially more atelectasis than the 40% oxygen group.
These two studies challenge the dogma we have been teaching for many years that 100% oxygen should be used for both induction and emergence from anesthesia.
For many years, I have practiced and taught the use of specific oxygen targets during induction, maintenance and emergence tailored to the patient’s needs. During induction and emergence, the goal is to use enough oxygen to increase the apnea time until hypoxemia occurs without promoting atelectasis. During maintenance, there are two goals – preventing atelectasis but also being able to detect untoward changes in oxygenation.
During induction and emergence, for most patients, there is a rationale to limit the FiO2 to 75-80% oxygen. This provides a much greater margin of safe apnea time than room air and, based upon available evidence, should include enough nitrogen to avoid absorption atelectasis. Are there times when I would choose to use 100% oxygen? Of course. During induction and emergence, for patients with a known oxygen dependence or where airway management may be especially challenging, 100% oxygen is warranted. Any resulting atelectasis can be managed intraoperatively with recruitment maneuvers and PEEP or postoperatively with non-invasive ventilation.
The next question is What is the optimal oxygen concentration during maintenance? The most common cause of an oxygenation problem during anesthesia is atelectasis and there are multiple potential causes in addition to the oxygen concentration used such as patient position, the use of muscle relaxants and the site of surgery. In addition to induction and emergence strategies for preventing atelectasis, it is important to be able to monitor for the occurrence of atelectasis and once detected, to ensure that a recruitment maneuver is effective. Atelectasis will cause a decrease in oxygenation typically detected by evaluating some index of the alveolar to arterial oxygen gradient like the PaO2 or SpO2 to FiO2 gradient. Atelectasis will also cause a reduced lung compliance. Both oxygenation and lung compliance can be assessed with bedside monitors. For brevity, this discussion will focus on oxygenation monitoring.
Pulse oximetry is the most commonly used tool for assessing oxygenation since arterial blood gas analysis is not routinely available. Here is the key point – Pulse Oximetry is insensitive to changes in oxygenation if enriched oxygen concentrations are used. Since the pulse oximeter measures percent saturation and not partial pressure of oxygen, it will be a late indicator of untoward changes in the A-a gradient if the inspired oxygen concentration is greater than 21-25%. The use of the SpO2 to FiO2 relationship to assess oxygenation and optimize ventilation is nicely described in a review article by Tusman et al. Advanced Uses of Pulse Oximetry for Monitoring Mechanically Ventilated Patients. Anesth Analg 2017:124, 62.
Unless the patient requires an enriched oxygen concentration to maintain the desired oxygen saturation, using no more than 25% oxygen will provide the conditions for rapidly detecting changes in oxygenation should they occur, and assessing the impact of a recruitment maneuver if needed to correct suspected atelectasis. There may be situations where a greater FiO2 is considered desirable to create more time to deal with hypoxemia should “something occur.” This may be a false sense of security since the greater FiO2 will only delay the ability to recognize that an oxygenation problem is occurring. That said, if enriched oxygen concentrations are used, it is useful to reduce the FiO2 to 25% or lower periodically, or certainly before emergence, and observe for a decrease in saturation to determine if a recruitment maneuver or other intervention is needed to correct atelectasis or another cause of oxygenation problems.
To summarize, the practice of using no more than 80% oxygen during induction and emergence for most patients has sound physiologic rationale and clinical evidence that it will prevent atelectasis. During maintenance, using an FiO2 less than 25% either continuously or periodically allows assessment of oxygenation using a pulse oximeter. Practicing with a specific oxygen target tailored to the needs of the patient is consistent with the desire to maintain physiologic homeostasis as close to normal as possible despite the trespasses of anesthesia and surgery.
The literature on atelectasis during anesthesia is extensive and worth a read if you are interested in the topic. There is a nice review specifically targeted at pediatric patients by Bruins, S., Sommerfield, D., Powers, N. & Ungern‐Sternberg, B. S. Atelectasis and lung recruitment in pediatric anesthesia: An educational review. Pediatr Anesth 2022:32,321.
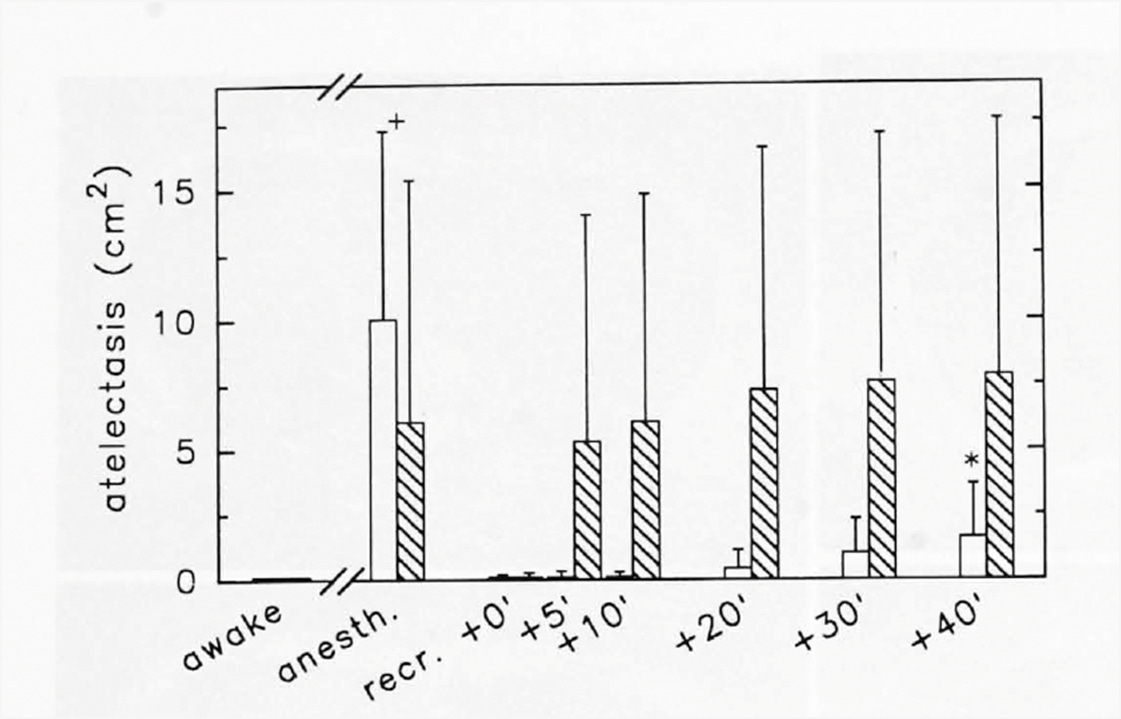
Figure from Rothen et al Anesthesiology 1995:82;832. Hashed bars are patients breathing 100% oxygen after recruitment. Open bars are 40%. Note rapid formation of atelectasis after just 5 minutes breathing 100% oxygen.
Rothen et al. Anesthesiology 1995:82;832.
From George D Politis, MD University of Virginia
I believe the recent PAAD here regarding tracheal intubation with deep anesthesia (TIDA) versus intubation with non-depolarizing muscle relaxants (NDMRs) might lead readers to wrong conclusions! I became interested in TIDA during my anesthesiology residency at UVA after Rosenburg and Gronert published a report in 1992 titled Intractable Cardiac Arrest in Children Given Succinylcholine and the FDA soon followed with a black box warning on succinylcholine. A common practice at that time, including for my instructor Fritz Berry, was to induce anesthesia with halothane, start an IV, administer succinylcholine, intubate the trachea (there were no LMAs), and administer an NDMR only if one was needed. A common practice after the black box warning was TIDA for children when procedures didn’t require NDMR, and that practice accelerated after the introduction of sevoflurane in the US around 1995. I’ve been a proponent of TIDA throughout my career, have designed and authored several clinical studies on the subject including one used in the Christensen et al metanalysis1 presented in the PAAD, and I’ve been an invited speaker on TIDA at several ASA meetings. I am familiar with many of the studies used in the metanalysis presented. I may have a blind spot with respect to this topic, but I have a wide field of vision.
Myron recently expressed a disapproving sentiment toward metanalyses. I share that sentiment to some degree, but I do appreciate metanalyses that combine studies with similar designs and outcome definitions and carefully import accurate data. Those metanalyses create strong evidence. I do have concerns about the accuracy and presentation of results by the Christensen et al metanalysis1 and the conclusions by PAAD reviewers. Their Table 1 shows they combined widely disparate studies: some look at TIDA with sevoflurane, some halothane, others propofol and remifentanil, some use N20 and others alfentanil, and some compared low-dose rocuronium to TIDA or to intubation with only 1-MAC of sevoflurane. It is hard to draw conclusions about any one TIDA technique when so many disparate techniques were included. Christensen et al1 used the primary outcome “difficult tracheal intubation” and used each authors definition of that outcome. In many, if not most of the studies incorporated in Christensen et al’s study, my study2 included, “difficult tracheal intubation” wasn’t defined at all. Most TIDA studies used the outcome “adequate intubation conditions” and often used the well-established Viby-Mogensen criteria3, or a modification, to define “adequate”. In my study2, the 5 patients with “inadequate conditions” that were translated in Christensen’s study into “difficult intubations” were not difficult intubations at all, at least not according to how an average anesthesiologist defines “difficult intubation”. Two of the 5 had inadequately abducted vocal cords, one developed laryngospasm, and two had an inadequate view on the initial DL but good subsequent view. We found no difference in inadequate conditions between the low-dose rocuronium group (0.25 mg/kg) and the deep halothane group in Honduran patients receiving cleft lip or palate surgeries. I could discuss other individual studies used by Christensen et al, but I suspect by now you understand that broad sweeping conclusions drawn from this metanalysis may be misleading.
One might say why bother with TIDA in the age of sugammadex? One reason is cost: an AI search I recently conducted with Google found that sugammadex costs US hospitals between $70-100 per 2-ml vial, which may become less within a year when it comes off patent but is likely to remain appreciable. Another reason is to avoid side effects of both NDMRs and sugammadex, which includes anaphylaxis, bradycardia, and residual neuromuscular block (until quantitative neuromuscular monitoring is provided by all hospitals and uniformly utilized). As an anesthesiologist involved in global health for the last 30 years who has practiced substantially in low resource countries (LRCs) in South and Central America, Africa, and Asia, I can assure you that sugammadex and quantitative neuromuscular monitoring are either not available or minimally available in LRCs, and those countries constitute a large part of the world. You are lucky in LRCs if you can find a standard twitch-monitor. NDMRs are often avoided in LRCs because both NDMRs and neostigmine are difficult to source, are deemed too costly especially when cost is transferred to impoverished patients. NDMRs often lead to prolonged neuromuscular block in LRCs because blockade is often not reversed and routine intubation with succinylcholine remains rightfully very much alive and a valuable tool when inexperienced trainees intubate. Fortunately, TIDA is quite safe in experienced hands when intubating healthy children, and deep anesthesia reliably produces similar intubating conditions to NDMRs and similar incidence of laryngospasm, when protocols are followed that allow us to know the amount of anesthesia that will reliably produce good or excellent intubation conditions4,5,6,7 (good or excellent = “adequate”)……which is a topic for another day.
Alex de Tocqueville, a French scholar who is best known for coming to America to study our celebrated and now challenged democracy, wrote two volumes called Democracy in America around 1840. He made many astute observations that hold true today. He spoke about the strong bandwagon effect in America. The day may come when anesthesiologists all around the world should get aboard the bandwagon calling for use of NDMRs for intubation of all pediatric patients, but I for one don’t believe that day is near.
References:
1. Christensen M, Creutzburg A, Vested M, et al. Effects of avoidance versus use of neuromuscular blocking agents for facilitation of tracheal intubation in children and infants. Eur J Anaesthesiol 2026: 43: 335-348.
2. Politis G, Brill J, Jones J. Use of low-dose rocuronium for intubation of children during volunteer surgery abroad. Pediatric Anesthesia 2005; 15: 648-52.
3. Viby-Mogensen J, Engbaek J, Eriksson L, et al. Good clinical research practice in pharmacodynamic studies of neuromuscular blocking agents. Acta Anaesthesiol Scand 1996: 40: 59-74.
4. Crawford M, Hayes J, Tan J. Dose-response of remifentanil for tracheal intubation in infants. Anesth Analg 2005; 100: 1599-604.
5. Politis G, Frankland M, James R, et al. Factors associated with successful tracheal intubation of children with sevoflurane and no muscle relaxant. Anesth Analg 2002: 95: 615-20.
6. Politis G, Stemland C, Balireddy R, et al. Propofol for pediatric tracheal intubation with dep anesthesia during sevoflurane induction: dose according to elapsed time for two age groups. J Clin Anesth 2014; 26: 25-35.
7. Lerman J, Houle T, Matthews B, et al. Propofol for tracheal intubation in children anesthetized with sevoflurane: a dose-response study. Pediatric Anesthesia 200-9; 19: 218-24.