More than 30 years ago, Chuck Berde and his colleagues published one of the first papers on the use of methadone as “a poor man’s alternative to IV PCA” in children.(Berde, C.B., et al., Comparison of morphine and methadone for prevention of postoperative pain in 3- to 7-year-old children. J.Pediatr., 1991. 119(1 ( Pt 1): p. 136-141) It completely changed my thinking and I became a “believer”. And you had to be a believer because there was and continues to be an enormous amount of resistance to using methadone from families, surgeons, colleagues, and hospital pharmacists. This PAAD first posted on August 21, 2021 and lays out the pharmacokinetic and pharmacodynamics of methadone in children when used as a component of a multi-modal pain management. In tomorrow’s PAAD we will review a recently published paper by Mok et al. which reports a single center’s retrospective ERAS study/protocol of how methadone can be used to reduce opioid use following pediatric (really adolescent) posterior spine fusion surgery. Myron Yaster MD
Original article
Sadhasivam S, Aruldhas BW, Packiasabapathy S, Overholser BR, Zhang P, Zang Y, Renschler JS, Fitzgerald RE, Quinney SK. A Novel Perioperative Multidose Methadone-Based Multimodal Analgesic Strategy in Children Achieved Safe and Low Analgesic Blood Methadone Levels Enabling Opioid-Sparing Sustained Analgesia With Minimal Adverse Effects. Anesth Analg. 2021 Aug 1;133(2):327-337. PMID: 33481403
Editorial
Elizabeth De Souza , T Anthony Anderson. Pediatric Perioperative Methadone Dosing Trial: An Illustration of the Challenges in Conducting High-Quality Pediatric Anesthesia Research. Anesth Analg. 2021 Aug 1;133(2):324-326. PMID: 34257193
Over the past 3 months, there have been 2 PAADs focusing on methadone (June 17, “Methadone + Ketamine 2+2 = 8”) and pediatric clinical drug research and FDA drug labeling (May 27, “Why are children always last”?). Topics that were and are of great interest to Ron Litman and me. Today’s PAAD on the pharmacokinetics and dynamics of methadone in adolescent patients undergoing posterior spine and pectus excavatum surgery and the accompanying editorial add more fuel for your thoughts on these topics.
Before beginning, I’ve got to admit that I’m biased. I think methadone is a fantastic drug, underutilized by many anesthesiologists. It is a long-acting, inexpensive opioid and the only mu-opioid agonist that is also an NMDA receptor antagonist. These properties make it a fabulous analgesic for acute perioperative pain, cancer pain, sickle cell vaso-occlusive crisis pain, and opioid use disorder. Its onset is as rapid as fentanyl’s but it has a longer duration of action. It reduces the need for short-acting opioids and lowers the risk for chronic postsurgical pain. I only wish methadone had a different name…there are so many anesthesiologists, surgeons, pharmacists, and families who only associate it with addiction, rather than with its many salutary effects. Unfortunately, based on my experiences in Colorado, many of you and your OR pharmacists may have little experience with using it or may even be afraid of using it because of the fear of respiratory depression or prolonged QT. This paper by Sadhasivam et al., goes a long way in answering the many questions on how to use this fantastic drug safely.
Methadone has a long half-life of elimination (t ½ beta) and is rapidly redistributed (short half-life of redistribution, t ½ alpha). Sooo, like fentanyl or propofol, a single dose will lose its effectiveness over a short period of time (usually 2-4 hours) because of rapid redistribution and the consequent fall in blood levels to subtherapeutic levels. Further, “it is bound by alpha-1 acid glycoprotein (AAG), an acute-phase protein with a half-life of 2–3 days; AAG increases by 2- to 7-fold in response to stressors such as surgery. This postoperative increase in AAG levels can potentially bind circulating methadone and decrease “free” methadone available for receptor binding and clinical analgesia.” This role of AAG in the context of perioperative methadone was previously unexplored. As the authors state: “This study aimed to characterize pharmacokinetics as well as safety and efficacy of multiple small doses of perioperative methadone in children—integrating the influence of AAG. Our hypothesis was that the multiple small perioperative dose methadone-based multimodal analgesia will result in safe blood methadone concentration (<100 ng/mL) (primary pharmacokinetic end point), as well as will reduce postoperative opioid use within 72 hours (primary clinical end point) and the risk of respiratory depression (secondary clinical end point) when compared to an historical study with single-dose methadone in children undergoing PSF”.
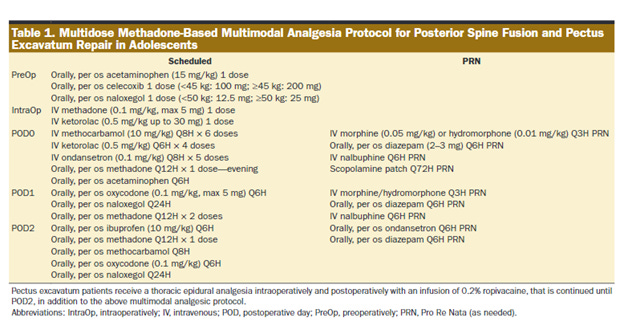
Before reviewing the results, this study utilized a very specific multimodal analgesic regimen. The methadone and other drug doses and the schedule of administration are in the table below. Note they utilized multiple, scheduled small IV and oral doses of methadone rather than a single large dose. Because of its great bioavailability the IV methadone dose=oral dose. It should be noted (and was so noted in the accompanying editorial) that there was no control group; rather the outcomes were compared to the results of an historical study of the use of single dose methadone for adolescents undergoing PSF.(1)
The key findings: Smaller scheduled doses, along with acetaminophen, nsaids, and naloxegol produced sustained analgesia, opioid sparing, and no respiratory depression or QT prolongation. The authors conclude: “novel multiple small perioperative methadone doses resulted in safe and lower blood methadone levels, <100 ng/mL, a threshold previously associated with respiratory depression if exceeded. This methadone dosing in a multimodal regimen resulted in lower blood methadone analgesia concentrations than the historically described minimum analgesic concentrations of methadone from an era before multimodal postoperative analgesia without postoperative respiratory depression and prolonged corrected QT (QTc).
The authors also found that AAG levels rose throughout the study directly affecting the amount of free methadone available for both methadone’s efficacy and detrimental effects. I’m not sure how we can make use of this information in actual “combat” situations. AAG and free methadone blood levels are not easily available, if they are available at all, in clinical practice. On the other hand, it might explain why some patients may need more or less drug in practice. Finally, in this study, breakthrough pain was treated with PRN IV morphine or hydromorphone. On postop days one and two, patients were scheduled to receive both oral methadone AND oral oxycodone. In my previous practice in Baltimore, we would often treat breakthrough pain and maintenance with only methadone.
The short t ½ alpha and long t1/2 beta, as well as the rapid rise of CMax following an IV dose, explains why in my own practice, unlike in this paper, I redose methadone early (around 3 hours) and then stretch out subsequent dosing. Obviously, with sustained dosing, methadone will have a prolonged duration of action. Certainly the excellent pain control with minimal adverse effects in this study was achieved not only as a result of the use of methadone, but due to the multimodal analgesia from the combination of adjuvant medications (NSAIDS, muscle relaxants (methocarbamol and valium), and naloxegol. Finally, methadone is incredibly inexpensive, making it a very cost-effective opioid analgesic in your practices.
There is much more information in this paper and in the editorial (which I didn’t review for you at all in this PAAD). So, if you have the time, I’d highly recommend reading them. And for those of you who have journal clubs, I think these would be great papers for discussion. Indeed, I’ve spoken to Sean Barnes, on the Hopkins faculty and a current SPA board member and social media expert, about setting up a monthly journal club on the SPA website based on some of the papers highlighted in the PAAD. Will keep you posted if this happens.
Myron Yaster, MD
References
1. Sharma A, Tallchief D, Blood J, Kim T, London A, Kharasch ED: Perioperative pharmacokinetics of methadone in adolescents. Anesthesiology 2011; 115: 1153-61